How heat moves from one substance to another
We now ask how it is that heat moves between substances. The 0th Law of Thermodynamics tells us that heat always moves from hotter substances to colder ones. Consider these situations:
- A cup of hot cocoa loses its heat to the cup and the table upon which it sits, and to the air.
- When you touch a cold object, it feels cold because heat is moving from your hand to the object.
- When you touch a hot object, it feels hot because heat is moving from the object into your hand.
- Did you ever notice that even on a warm day, a piece of metal at room temperature will feel cool?
- If you walk outside on a cold, calm day, you will lose body heat, but not nearly as fast as if there is wind at the same temperature.
Our goal in this section will be to understand all of these phenomena in terms of how heat moves from one thing to another.
Consider the figure below. The Thinker loses his body heat in four main ways: by conduction, radiation, convection and evaporation.

1. Conduction
Conduction of heat is transfer through physical contact. In hotter objects, there is more atomic and molecular motion. Atoms and molecules "jiggle" faster and more wildly. When a hot object touches a colder object, its surface atoms and molecules bang into those of the cold object, causing them to jiggle faster — they heat up. This will continue until both objects are the same temperature, or are in thermal equilibrium, one having lost some heat and one having gained an equal amount. At thermal equilibrium, the heat flow rate is the same in each direction.
Conduction is one of the fastest ways to transfer heat. When you're camping, for example, you can become hypothermic at night - even on a warm night, by sleeping directly on the ground. The ground temperature is usually quite a bit cooler than your body temperature, therefore heat will flow from you to the ground. That's why an essential piece of camping gear is some sort of sleeping pad to insulate you from the ground. But what does it mean to insulate?
Insulation
A thermal insulator is a material through which heat does not transfer easily. Conduction relies on direct transmission of the atomic jiggling that is heat. If the atoms of a substance are tightly packed, then that jiggling is efficiently transmitted from one atom/molecule to another across it.
Such a substance is a good conductor of heat, and a poor insulator. See the left panel of the figure below. An extreme example of this kind of substance is diamond. Diamond is a nearly perfect tetrahedral lattice of carbon atoms. When one atom vibrates, others in the fully interconnected lattice "feel" it nearly instantaneously and transmit the vibration to the next atom in the line.
A better situation would be the same kind of order, but with more open space. Now the jiggling has to be transmitted over a more convoluted path with more branch points into which it can split and dissipate (2nd frame below). Plastics and wood are rough examples of this kind of insulator.
Even better are substances with few connections between molecules. This might be the case for something like pink fiberglass insulation, which is made of long, thin glass fibers spun in such a way that there is a tremendous amount of empty space between them.
The best insulator (below right) would be a vacuum or near vacuum, where most of the atoms/molecules have been removed (there's one left). If there's nothing to transmit jiggling from one substance to another, it won't be transmitted. Vacuum thermos bottles work on this principle.
Conduction
The words conductor and insulator are used in two different ways in physics. Materials may conduct electricity well and they might be good thermal conductors. Generally, things like metals that conduct electricity are also good conductors of heat. Electrical insulators do not conduct electricity well. Most ceramics and Teflon™ are good examples. Materials that don't allow the flow of heat are called thermal insulators. Often these contain a lot of "dead air" space, like solidified foams.

2. Radiation
We have established that heat is just the internal energy of the movement of atoms and molecules in a substance. When objects absorb heat, molecules vibrate with higher kinetic energy (faster). Once in a while, a molecule "relaxes" to a state of lower vibrational energy. Energy cannot simply disappear, so the lost energy has to go somewhere. It is radiated out into the world, generally as infrared light, a kind of electromagnetic radiation.
The hotter an object, the more energy it radiates. Think of a red-hot piece of metal. As the temperature of an object increases, the frequency of electromagnetic radiation that it emits shifts upward. Higher frequency (and shorter wavelength) means higher energy.
Very hot objects glow red, then blue, then white as the temperature increases. This phenomenon is known as blackbody radiation.

Photo: Fir0002/Flagstaffotos (Wikipedia Commons)
An object doesn't need to glow with visible light to radiate heat. You give off heat just sitting there. In fact, a human being is in thermal equilibrium with her/his surroundings at about 72˚F.
That means your basic metabolism pumps out enough heat to raise you to normal body temperature, 98.6˚F just by virtue of you being alive.
Examples of radiative heat loss are all around. If you don't have baseboard radiators in your house, at least you've probably seen them (right). They are built to maximize the surface area from which heat can escape. Heated water in a metal pipe warms attached metal fins, which provide ample surface area from which radiated heat can escape.
Heat radiates from any object, provided the environment around it is cooler. A great deal of heat radiates from the human head in cold weather, which is why wearing a hat can go a long way toward keeping you warm.
Wearing insulating clothing can help to keep radiated heat trapped next to the skin. The function of warm clothing is to keep radiated heat in, keep wind out (see convection below) and to provide an insulating hedge against conductive cooling.

The speed of heat transfer: Thermal gradients
The larger the difference in temperature between two objects, the faster heat will transfer from the hotter one to the cooler one.
The difference in temperature between two objects is called a thermal gradient (gradient = slope). The steeper the thermal gradient, the faster the flow of heat. For example, if a hot object is placed into a container of warm water, the hot object will lose heat to the water at some rate. But if the same object is placed in ice-cold water, not only will more heat transfer, it will transfer more rapidly, in proportion to the difference in temperature (which of course is changing).

The rate of heat transfer between two objects is proportional to the temperature gradient and to the amount of surface area through which the conduction occurs. The figure below shows the heat transfer as the temperature of a hotter object transfers heat to a cooler one. We assume, for the graph, that the mass of the hotter object is very small compared to the cooler object, so that the temperature of the cooler object doesn't change appreciably - it is an "infinite heat reservoir."

Of course, as the temperature difference between two substances decreases, the rate of heat transfer slows down. This leads to the curved graph on the right. The slope of the curve is steep when the temperature difference is large (fast heat transfer), but less steep as the difference decreases (slower heat transfer).
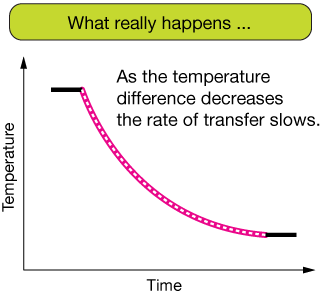
The equations that model such heat transfer are a product of differential calculus, so I won't discuss them here. See, for example, Newton's Law of Cooling.
3. Convection
Take another look at our Thinker. Without any air movement, the body radiates heat – like it always does. Over time in still air, a "blanket" of heat will form around the body as radiated heat warms air molecules. Now the temperature of the air just above the body is warmer than the ambient temperature, so the thermal gradient is less steep and heat loss slows down.
But turn on some wind and blow those layers of heated air away and we end up with the steepest possible thermal gradient: ~37˚C (98˚F) skin right next to colder air.
A sure way to ensure the fastest possible loss of heat is to quickly blow away any air warmed by radiation from the body. Removing the heated air maintains the steepest possible thermal gradient, and therefore the fastest possible heat loss.
That's why freezing days can feel quite different depending on whether there is wind. This real phenomenon is what led to development of the wind-chill concept, a scale to remind us that low temperatures will feel colder in proportion to the speed of the wind.

4. Evaporation
We know from our study of heat and the section on water that the latent heat of vaporization, ΔHv, of water is large. It takes a significant amount of energy to convert water (or any liquid, but especially water) from the liquid phase to the gas phase.
Evaporation can thus be an effective means of removing heat from something, like a body. Humans sweat to reduce heat. Our skin cells pump water to the surface, our body heat evaporates it, and it carries that heat away into the atmosphere.
If you live in a very humid area, you won't be able to cool yourself as efficiently in the summer, because sweat won't evaporate from your skin so readily. That's because the air can be nearly saturated already with water vapor.
Dogs and other mammals pant, evaporating water with heat from their breath to cool.
Isopropyl alcohol (rubbing alcohol) is used to simulate a cooling effect on the skin. Most alcohols evaporate at temperatures below human body temperature (98.6˚F = 37˚C), so they evaporate quickly from the skin, taking heat with them into the gas phase.

![]()
xaktly.com by Dr. Jeff Cruzan is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License. © 2012, Jeff Cruzan. All text and images on this website not specifically attributed to another source were created by me and I reserve all rights as to their use. Any opinions expressed on this website are entirely mine, and do not necessarily reflect the views of any of my employers. Please feel free to send any questions or comments to jeff.cruzan@verizon.net.