Intermolecular forces

Why does water bead up like this on certain surfaces, like a waxed car or glass? Why doesn't it wet the entire surface evenly? The answer is that the forces of attraction between water molecules – intermolecular forces – are stronger than the attractive forces between the water molecules and the surface.
Intermolecular forces are the glue that hold many materials together. They give many substances their properties, such as melting and boiling temperatures.
When we talk about ionic or covalent bonding, we're talking about attractive forces that are relatively large.
These bonds are on the order of 100 times stronger than the bonds that result from the attractive forces we'll discuss in this section. Nevertheless, differences in the forces between atoms and molecules can lead to profound differences even between compounds that would seem similar.
All of the intermolecular forces we'll talk about in this section arise from the fundamental arrangement of the electrons in atoms and molecules.
The type and strength of an intermolecular attraction or repulsion depends on how many electrons are present, how they are arranged in bonds, what kinds of bonds are present, and so on.
In this section, we will discuss five fundamental kinds of intermolecular forces. As you work through these, try to keep in mind their similarities more than their differences.
There are six types of intermolecular forces
- Electrostatic or "Coulomb" forces
- Multipole forces, such as the dipole—dipole force
- Induction forces
- The dispersion force, or "London dispersion"
- Hydrogen bonding
- The hydrophobic effect
The electrical force between charged particles (atomic or molecular ions, protons or electrons) is one of the four fundamental kinds of forces in the universe (the others are gravity and the strong and weak nuclear forces). We call this the electrostatic force.
We describe these forces using Coulomb's law. The Coulomb or electrostatic force is the strongest of the intermolecular forces; it accounts, among other things, for the ionic bonding of salts, such as NaCl.

For oppositely-charged atoms, the electrostatic force is attractive, but that's an oversimplification of what really happens between atoms.
All atoms are surrounded by negatively-charged electrons, so at very close range, when the electrons come into close contact, they actually begin to repel one-another. This repulsive force is what causes objects not to fall right through objects upon which they sit, e.g. a lamp on a table. It's the repulsion between electrons that keep objects from actually "touching".
In the graph below, the black line is the electrostatic force for two oppositely-charged ions. It shows (unrealistic) infinite attraction at small distances, $r$. The red curve incorporates the added repulsion at short distances, for a more realistic view of the force at work between charges.
Any chemical or physical bond (bond due to IMFs) occurs because its formation reduces the potential energy of the particles involved, and is a balance between attractive forces and electrostatic repulsion between electron clouds.
A typical potential energy vs. separation curve for the formation of a bond
Think for a moment about an inert gas atom, say Argon (Ar). Although we know that the electrons aren't arranged in a strictly spherical cloud around the nucleus (p-orbitals and all that), because the atom is free to rotate, the distribution of electrons is more-or-less spherical.
Because an atom is spherically symmetric, we can't identify any one "side" or portion of its electron cloud that is any more or less positive or negative than any other.
Now think of a molecule like hydrogen chloride (HCl). It doesn't have that spherical symmetry. It's a sphere with a "bump".
In fact, because the chlorine atom is quite electronegative, it pulls the single H-atom electron mostly away from the hydrogen, leaving a mostly bare proton. HCl has two different "ends", one clearly more positive than the other.
The H-atom side is more positive than the Cl-atom side (see the picture below).
This situation is sometimes described with this language: An atom is isotropic. It looks the same viewed from any direction. The HCl molecule is anisotropic (not isotropic) because as we approach it from different directions, we get a different view. Space is isotropic. It has no special direction. We only define "up" as north on our globe out of long-standing convention, not because space has any special up or down direction.

Isotropic / anisotropic
A thing that is isotropic looks the same from any direction. It has no special oreientation, like a uniformly-colored sphere.
A thing that is anisotropic has one or more distinguishable orientations. A pencil is anisotropic because it has two distinguishable ends, but (aside from the labeling) it is isotropic with respect to rotation about its long axis.
Permanent dipoles
You can see how the symmetry of a molecule, or we often say the "breaking" of some underlying symmetry, can lead to an asymmetric distribution of the electrons around it.
This makes one part of the molecule "less negative" (and therefore more positive) than another. A molecule that clearly has two sides or ends, one more negative than the other, is said to possess a permanent dipole (two poles, like a battery, + and -), and has interesting properties because of it. The linear molecule HCl is a good example (below).

Non-linear molecules can have permanent dipoles, too. In the top row of the table below are some molecules which are highly symmetric, and because of that symmetry, cannot have permanent dipoles.
For example:
- Borane is a flat (planar) molecule with three equivalent bond angles. It's not possible to find a unique "end". If we substitute a bromine for one of the hydrogens to make bromo borane (BrBH2), we break the symmetry to form a dipole.
- Methane as a 3-D analog of borane. It's tetrahedral structure, with four equivalent bond angles gives it too much symmetry to be a dipole. But if we substitute a chlorine for one of the hydrogens to make methyl chloride, that symmetry is broken and the Cl end is more negative than the rest of the molecule.
- The benzene molecule (C6H6) has six-fold symmetry and is planar. If we put an OH onto one of the carbons, we form polar phenol (C6H5OH).
- Finally, carbon dioxide (CO2) is a linear molecule with identical ends. Water has identical "ends" but it's bent, with lots of negative electron density near the oxygen. Water is actually very polar.

Dipole strength: The dipole moment
We need a way to measure the relative strengths of dipoles, because a dipole can exert a force on another atom or molecule, and the amount of force exerted will depend on the strength or magnitude of the dipole.
The strength of a dipole depends on how much charge difference exists between one "end" of a molecule and another, and the separation of those ends. We call the measure of dipole strength the dipole moment. In mathematics, a moment is a (vague) measure of shape.
This table lists the dipole moments of some commonly encountered substances. The unit of measurement of dipole moment is the Debye (D).
| Compound | $\mu \; (D)$ |
|---|---|
| Water (H2O) | $1.85$ |
| Water (H2O) | $1.85$ |
| BrCl | $0.6$ |
| HI | $0.44$ |
| HF | $1.82$ |
| Ammonia (NH3) | $1.42$ |
| Chloromethane (CH3Cl) | $1.8$ |
| Ethanol (C2H5OH) | $2.69$ |
| Carbon dioxide (CO2) | $0$ |
Practice problems — 1
Predict whether each molecule below is polar (has a dipole moment) or non-polar. You might need to sketch out a Lewis structure to tell.










Which of the following liquids might you expect to have the highest and lowest boiling points? Explain your responses.

How Dipoles Interact
The figures on the right show how two or more dipoles interact when they are close. First, we introduce a shorthand notation, the crossed-arrow symbol; the plus goes on the positive side of the dipole and the arrow on the negative side.
When dipoles are free to rotate, they tend to class, as shown. This happens because of the twisting force shown. That twisting force is simply due to the electrostatic force: A region of high negative charge will repel the more negative end of a dipole and (relatively speaking) attract the more positive end.
When many dipoles collect, e.g. in a solid, they tend to class as shown in the bottom panel. Note the each + and - charge is surrounded by opposite charges.
A note on + and - : δ+ and δ-
When designating the + and - ends of a molecular dipole, we generally use the symbols δ+ and δ- rather than + and -. We use the Greek letter δ (delta) to indicate that we're not talking about a full positive or negative charge, just a partial charge. δ+ means "a little more positive."

The Greek alphabet
| alpha | Α | α |
| beta | Β | β |
| gamma | Γ | γ |
| delta | Δ | δ |
| epsilon | Ε | ε |
| zeta | Ζ | ζ |
| eta | Η | η |
| theta | Θ | θ |
| iota | Ι | ι |
| kappa | Κ | κ |
| lambda | Λ | λ |
| mu | Μ | μ |
| nu | Ν | ν |
| xi | Ξ | ξ |
| omicron | Ο | ο |
| pi | Π | π |
| rho | Ρ | ρ |
| sigma | Σ | σ |
| tau | Τ | τ |
| upsilon | Υ | υ |
| phi | Φ | φ |
| chi | Χ | χ |
| psi | Ψ | ψ |
| omega | Ω | ω |
Higher multipole moments
It turns out that the dipole moment is not the only kind of charge moment a molecule can have. CO2 has no dipole moment, but it does have a quadrupole moment. While these higher moments must be considered in some weak interactions, they are of less importance than the dipole.
At a meeting of the Faraday Society in England years ago, a Harvard Professor, responding to a point made in a discussion of weak bonding between molecules said, "Aha! The quadrupole moment ... last refuge of a scoundrel!" Nerdy that I remember that.
We have seen that charges and regions of relative charge difference can interact to attract and/or repel one another. How does a non-charged, non-polar atom or molecule interact with a dipole? It turns out that a dipole can induce a dipole moment in, say, an atom. Play the animation below to see how it works.
The animation simulates the distortion of an electron cloud as a permanent dipole (HCl, in this case) is brought near a neutral atom, the anisotropy of its charge distribution redistributes the electron density of the atom. Electrons are attracted away from the atomic nucleus and toward the positive end of the dipole. An atom is spherically symmetric, so it can't have a permanent dipole, but this distortion disrupts that symmetry and induces a small, temporary dipole moment in it.
This effect is stronger with the strength of the permanent dipole, and with the polarizability of the atom.
The induced dipole interacts with any permanent dipole just as a permanent one would, induced dipoles are just weaker, so any attraction or repulsion is weaker.
Any polar molecule can similarly rearrange the electron cloud of any molecule, including molecules that already have permanent dipole moments. In fact, the induction force accounts for about 10% of the total cohesive binding force of liquid water - the force that accounts for the beading of water on a surface, the first image of this section.
Polarizability
An atom or molecule is polarizable if its electron cloud can be distorted in the presence of an electric field, like the one that might be experienced by being near a permanent dipole.
Polarizability increases with the number of electron shells, so it increases down groups (columns) on the periodic table.
The dispersion force, also known as London dispersion, is a purely quantum-mechanical force, and can only be truly understood by applying the laws (and mathematics) of quantum mechanics to the outer electrons of atoms.
It goes something like this: Think about two inert-gas atoms sitting side by side. From time to time in an atom the electron density is momentarily unbalanced, momentarily spoiling the spherical symmetry and forming a small dipole. This moment can induce a similar fluctuation in the other atom, and so on. Such fluctuations can become correlated (they occur more or less in sync.), producing an overall attraction between atoms that we might otherwise think would have no mechanism of attraction. That's roughly what the dispersion force is.
This animation illustrates how such a correlation of electron-cloud asymmetry might look. It's highly stylized, just a cartoon. A little funny really, atoms doing a jiggle dance.
The dispersion force scales roughly with the number of electrons for atoms, and similarly for molecules—the bigger the atoms the larger the polarizability, and the larger the attractive force due to the dispersion effect.
Measuring the effect of the dispersion force between atoms
We know that the dispersion force is real by looking at the properties of inert gases, where the only possible intermolecular force is dispersion.
This graph shows that helium (He), with only two electrons, has a much lower boiling point (the temperature at which atoms are liberated from the liquid) than larger inert gas atoms like Krypton (Kr) and Xenon (Xe). As the dispersion force becomes larger, it takes more energy to liberate an inert gas atom from the liquid because it is stuck to its neighbors more strongly.
Note that the dispersion attraction doesn't scale exactly with the atomic number - the graph is not linear. That means that dispersion must scale with more than just the number of electrons.

Hydrogen bonding is a very important kind of intermolecular interaction in biological chemistry. It is a prevalent force in water and any reaction that takes place in it; it is the force that holds the two strands of the DNA helix together; and it is the most important force determining the structure of protein molecules.
Hydrogen is the most abundant element in the universe. It is ubiquitous in the molecules of life: most are hydrocarbons, and of course water is H2O.
When hydrogen is bonded to an electron-withdrawing element, especially oxygen, it is a good candidate for H-bonding.
The essence of H-bonding is the attraction between an essentially bare proton (because it is attached to an electron-withdrawing atom) and an electron-rich atom, such as oxygen, which usually bonds in such a way as to expose two lone electron pairs.
The diagram below shows a H-bond between two water molecules.

The H-bonds between the bases of DNA are shown in the figure below. AT pairs are held together by two H-bonds and GC pairs by three.
Similar H-bonds hold the bases of RNA together to give RNA molecules their unique structures.
The alpha-helices and beta sheets, as well as other structural elements of proteins are held in place by H-bonds.
Which atoms can H-bond?
In many texts, the only atoms bonded to a hydrogen that can H-bond are nitrogen (N), oxygen (O) and fluorine (F). These atoms are small and quite electronegative, so they produce a large bond dipole, in which the electron of the H-atom is pulled tightly to the larger atom. The result is an exposed + charge at the hydrogen location.
O—H N—H F—H
We ought not to limit H-bonding to these atoms, however. It is possible to have weak H-bonding between S-H and P-H compounds as well. There is a continuum of bond-dipole strengths between small atoms and hydrogen.
In the biological context, H-bonding of N-H and O-H compounds is by far the most important, but S-H and P-H hydrogen bonds, though weaker, may still contribute significantly to the properties of biological macromolecules.

Finally, there is one more important effect that originates from forces between molecules, the hydrophobic effect. Substances like oils, fats and waxes that do not dissolve in water exhibit the hydrophobic effect. In terms of the energy of forming a solution, it is more energetically favorable for these non-polar substances to bunch together rather than be surrounded by polar water molecules, like this:

What generally happens is that non-polar molecules bind weakly to one another, thus reducing the amount of surface they present to water.

Roll over or tap the image below to see how this works on a larger scale. Hydrophobic molecules clump together, burying hydrophobic surface area within the non-polar mass. In this way the amount of interaction between non-polar solute and polar solvent (water in this case) is minimized.

Some non-polar substances, like olive oil, will actually completely phase-separate from water, floating on top (if the oil is of lower density than water) or sinking to the bottom. In this way, the water-nonpolar interactions are minimized to just the interface between the two layers of liquid.
The hydrophobic effect is very important in all kinds of chemistry. It has a profound effect on the structure of proteins, long chains of amino acids, about half of which bear non-polar side chains, and the other polar side chains. Proteins function in aqueous solution inside cells, so their 3-D structures are partly determined by the energetic drive to bury non-polar amino acid side chains inside the protein mass, away from surface water molecules.
hydrophobic
Literally hydro = water, phobic = fearing → water-fearing. Oily, fatty or waxy substances that do not dissolve in water are hydrophobic. The floating of oil on top of water is an example of the hydrophobic effect.
The boiling point of normal alkanes (CnH2n+2) increases with number of carbons
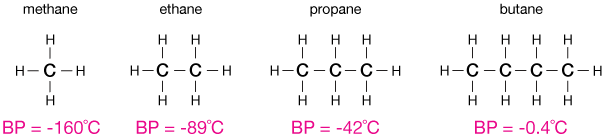
The figure above shows the increase in boiling point of normal alkanes (carbon-hydrogen compounds with only single bonds), as the number of carbons is increased. In fact, the trend continues, as shown in the graph below.

An increase in boiling temperature reflects the difficulty of liberating an alkane from the bulk liquid. Larger alkanes bind more tightly to their hydrophobic liquid phase, and are therefore more difficult to remove. That is, it takes more energy in the form of heat (which we measure with temperature) for a molecule to fly off into the gas phase.
One argument for this trend is that longer alkanes have more hydrophobic surface area to share with neighboring alkanes. The stability gained by burying this large amount of surface area is greater than the instability incurred by being surrounded by a polar solvent like water.
On the other hand, branched alkanes — alkanes with alkane (or aliphatic) side chains — have lower boiling points than their linear counterparts. Here are two examples. Notice that the BP of isobutane is lower than that of butane (figure above). These molecules have lower surface area then their linear alkane versions, which is consistent with our theory.

Here are more realistic models of neopentane (C5H12). On the left is a space-filling model, which is a decent representation of where the valence electrons are in a molecule.

Practice problems — 2
For the following pairs of molecules or atoms, decide which intermolecular forces are present, and which makes the biggest contribution to the cohesive force between them.
-
Ne & Xe
Answer
The only possibility for cohesion between Xe and Ne is the dispersion force. Xe is quite polarizable and Ne is less so. Xe-Ar and Xe-Xe would bond more strongly. The boiling point of Ne is 27K, while the boiling point of Xe is 166K.
-
CO2 & CO2
Answer
O=C=O has no dipole moment, so the dispersion force is the dominant cohesive force. CO2 does have a quadrupole moment which does contribute to its cohesion. You might think of a quadrupole moment as two dipoles, back-to-back, and pointing in opposite directions.
-
CO2 & Ar
Answer
O=C=O has no dipole moment, therefore it can't induce a dipole in the fairly polarizable Ar. The only IMF left is dispersion. Sometimes you have to think of it that way: What is/are the only force(s) that can account for the observations?
-
NH3 & Kr
Answer
Ammonia (NH3) has a permanent dipole moment, and can therefore induce a moment in Ar by distorting its electron cloud. Induction and dispersion forces form the intermolecular bond between these two.
-
NH3 & NH3
Answer
Ammonia (NH3) has a permanent dipole moment, so the dipole dipole interaction is important. The hydrogens of ammonia can H-bond to the lone pair of an adjacent nitrogen. This H-bonding force is stronger than the dipole-dipole force.
-
CH4 & C2H6
Answer
These are both hydrophobic molecules with no dipoles and no significant polarizability. They will mix well as gases or liquids if cold enough (like dissolves like), and they would separate into globules or layers in a polar solvent, like mixing an oil and water.
-
C2H5OH & H2O
Answer
Ethanol (C2H5OH) is polar, and dissolves readily in water (like dissolves like). They share dipole-dipole forces and hydrogen bonds (the stronger of the two) when mixed. Think of ethanol as a water molecule with one H substituted for a H3C-CH2—
-
H2O & H2O
Answer
Water has a permanent dipole moment (large for its size), so the dipole-dipole force is an important component of the cohesiveness of water. The largest contribution, however, comes from hydrogen bonding.
-
N2 & SO2
Answer
SO2 has a permanent dipole, and thus can induce a dipole in nonpolar N2. The dispersion force is also at work in this complex.
-
LiCl & H2O
Answer
Lithium chloride is a soluble salt, thus it dissolves in liquid water. Water molecules surround Li+ and Cl- ions in solution. The positive ends of water molecules tend to orient toward Cl- ions, and the negative ends toward Li+ ions.
-
C8H14 & H2O
Answer
C8H14 (octane) is a hydrophobic liquid. When mixed with water, it will float on top (it has a lower density) with a clear boundary between the two liquids. This minimizes the interface between these dissimilar (nonpolar - polar) liquids.
-
C8H14 & C24H50
Answer
Both of these substances are hydrophobic. They will form a solution at a high-enough temperature (like dissolves like). C24H50 (called tetracosane) is a waxy solid at room temperature and is insoluble in water.
-
SF6 & Xe
Answer
SF6 has octahedral symmetry, and thus no dipole moment. It basically looks like a large inert gas atom, and is often used to achieve that purpose. The dispersion force can hold these two gases together at low temperature.
-
HCl & Ar
Answer
Argon is polarizable, and thus the permanent dipole of HCl can induce a dipole in its electron cloud. The induction force is the strongest cohesive force between these particles, but dispersion is also a factor.
Consider the liquid phases of the following sets of three compounds. Order the compounds by increasing boiling point. Explain your ordering in terms of intermolecular forces.
-
Xe, Ne, Ar
Answer
The only IMF holding these liquids together is the dispersion force, which scales with polarizability, which, in turn, scales with atomic size, so the boiling points, from lowest to highest, should be Ne (-246˚C), Ar (-185˚C), Xe (-108˚C).
-
I2, Br2, Cl2
Answer
Aside from small quadrupole moments, the dominant force holding these liquids in the liquid state is the dispersion force, which scales with atomic size. From lowest to highest BP: Cl2 (-34˚C), Br2 (59˚C), I2 (184˚C).
-
C3H12
(cyclohexane), C6H6
(benzene), C6H5OH
(phenol)Answer
Cyclohexane and benzene are cyclic hydrocarbons that differ in the number of hydrogens, and benzene has a conjugated system of alternating single and double bonds that produces significant electron density on either flat side of the ring. Both are non polar and not terribly polarizable. Phenol is similar, but polar due to the OH group. It can even H-bond. We'd expect it to have the highest BP. Without more information, it's difficult to distinguish the BPs of the other two. They are: benzene (80˚C), cyclohexane (81˚C), phenol (182˚C).
-
CH3CH2CH3, CH3-O-CH3, CH3CH-OH
Answer
The alkane C3CH2CH3 (propane) is a non polar hydrocarbon with little in the way of cohesive force. It should have the lowest BP. CH3-O-CH3 is methyl-methyl ether, a polar molecule with dipole-dipole interactions. CH3CH-OH is ethanol, a polar molecule that can also hydrogen bond. From lowest to highest BP we have propane (-42˚C), dimethyl ether (-23˚C), ethanol (78˚C).
Indicate the type of intra molecular bonding that holds each of these compounds together, then consider what types of intermolecular forces would exist between molecules of the same kind, e.g. between CO2 molecules or between H2O molecules.
-
NaCl
Answer
NaCl is an ionic compound composed of Na+ and Cl- ions. The solid is held together by the electrostatic force. When dissolved in water, the ions are surrounded by dipoles of water molecules oriented to match the charge of the ion.
-
CO2
Answer
CO2 is held together by covalent (double) bonds. It has no dipole moment, so the principle source of intermolecular forces in the solid or liquid is the dispersion force, mostly through the electron-rich oxygen atoms. CO2 does have a small quadrupole moment which contributes to its intermolecular bonding.
-

Answer
-
I2
Answer
-
H2O
Answer
-

Answer
-
Rank these compounds in order of increasing boiling point. Remember that the greater the intermolecular cohesive (attractive) forces, the higher we expect the boiling point to be.

Answer
Pentane (C5H12) and isopentane (same chemical formula but different structure — a structural isomer) are hydrocarbons. They have small attractive intermolecular forces, and thus should have the lowest BPs. Branched alkanes (alkanes are CH compounds that contain only single bonds) like isopentane tend to have slightly higher BPs than unbranced ones, so at the bottom of our list are D, E. Cyclohexanone (C6H10O) is polar, but not as polar as acetic acid. Next on the list will be C,B. Finally, sodium acetate (CH3COONa), is an ionic compound. Its liquid state will be held together by the electrostatic force, which is stronger than the dipole-dipole force. In order of increasing BP, we have D, E, C, B, A. Isopentane (28˚C), n-pentane (36˚C), acetic acid (118˚C), sodium acetate (881˚C).
-
What causes C28H58 to be a non polar solid at room temperature ?
Answer
C28H58 is an alkane, a carbon-hydrogen compound with only single bonds. Alkanes are all hydrophobic, and have very small intermolecular attractive forces, due to dispersion. Small alkanes like ethane (C2H6) are gases at even very low temperatures. As we increase the number of CH2s in the chain, the attractive forces grow, so that larger alkanes like hexane (C6H6) are liquids, an larger ones like 28H58 are waxy hydrophobic solids that will float on water.
-
Which of these compounds might you expect to be soluble in water, and why?

Answer
We expect ionic (electrostatically bonded) compounds to be soluble in water, except for certain salts (see the solubility rules). Sodium acetate is an ionic compound, and all compounds containing Na+ are soluble. Ethanol is polar and can H-bond, so it's quite soluble in water. Br2 is nonpolar and covalently bonded. It is only slightly soluble in water, but it dissolves easily in nonpolar solvents like hexane or benzene (like dissolves like). Isopropyl amine is polar and can H-bond, so it should be water-soluble. Finally, cyclohexanone is soluble in water because it is polar. Because of the large hydrophobic portion of cyclohexanone, it's also soluble in many nonpolar solvents.
-
Which compound might you expect to evaporate most rapidly at room temperature: (a) C8H18 (b) C8H17OH (c) C8H17NH2 (d) C7H15COOH (e) C6H14 ?
Answer
The compound with the smallest cohesive (attractive) intermolecular forces will evaporate most rapidly. The polar compounds (b, c, d) are more cohesive than the alkanes (C-H compounds with only single bonds). Of those, the smaller alkane will have the smallest cohesive force, and that's C6H14 (hexane). Hexane does evaporate quickly at room temperature, but octane (C8H18) wouldn't be far behind.

![]()
xaktly.com by Dr. Jeff Cruzan is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License. © 2012-2025, Jeff Cruzan. All text and images on this website not specifically attributed to another source were created by me and I reserve all rights as to their use. Any opinions expressed on this website are entirely mine, and do not necessarily reflect the views of any of my employers. Please feel free to send any questions or comments to jeff.cruzan@verizon.net.